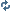|
Hi guys, as a continuation from the last post, I firstly want to thank you all for helping me out. But apparently due to the inability of my chemistry teacher to teach, i have a few additional questions that i hope you guys can help you answer.
Right now I'm studying electron configuration, and learning how to write elements using the 4 quantum numbers: n, L, ml, and ms. Plz refer to the below link for a moment:
http://images.google.ca/imgres?imgurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads_files/image002.jpg&imgrefurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads.htm&h=412&w=609&sz=17&hl=zh-CN&start=1&um=1&usg=__3F_i4ommegoDLULRa80xkMvvEeQ=&tbnid=tIokQ6UbR59zTM:&tbnh=92&tbnw=136&prev=/images?q=spdf,+shape+of+orbital&ndsp=18&um=1&hl=zh-CN&sa=N
The shape of the orbitals of electrons are s, p, d, f, and so on. I know that at the first energy level, n = 1, thus L = 0, which means the shape of the orbital is "s" capable of holding 2 electrons in a spherical shape.
When n = 2, L = 0 and 1, which means the orbitals at that energy level are in the shape of "s" and also "p". "s" can hold 2 electrons while "p" can hold 6, for a total of 8. But here comes my first question: do the orbitals "s" and "p" BOTH exist at the second energy level when n = 2, while the orbital at the first energy level "s" is separate from the orbitals in the second energy level?
*In addition, in my chemistry notes the terms px, py, pz are also used. Do all three terms represent ONE "p" orbital?
When n = 3, L = 0, 1, and 2. Which means the orbitals are "s", "p", and "d". "s" and "p" together can hold a total of 8 electrons. However, "d" supposedly can hold 10 electrons, and according to the link, the orbital "d" include 5 different orientations based on different x-y-z axises. My second question is, do all FIVE shapes exist together as the energy level "d"? If so, a total of 10 electrons in 5 orientations mean that each orientation can hold 2 electrons. How come there are 4 circles in each orientation then?
Lastly, ml is the magnetic quantum number, which can equal to -L .... 0 ..... + L, and it tells you the spatial orientation of the orbital, whether its along the x-axis, y- axis, or z-axis. Which value of ml corresponds with each orientation??
When it comes to the ms, which is the spin quantum number that tells the direction of spin of the electrons, for each orientation, DO I have to assign the positive 1/2 for the first electron first? Or does it not matter?
Thank you for your time.
    
|
Roffles

Pitcairn19291 Posts
It's been a while since i've taken Chem, but from what I can remember, SPDF equates to 2, 6, 10, 14 electrons respectively. px, py, pz are used to represent one p orbital that is correct if I'm not mistaken. As for quantum numbers, I'm not quite familiar, but I do know when it comes to spin, each electron can be assigned positive or negative. Usually, you're gonna fill one direction before the other, but which you assign is relatively negligible.
I believe p and s both exist because take for example the element Carbon, number 6 on periodic table. Electron configuration for carbon goes 1s2 2s2 2p2, which contains both p and s configurations. Correct me someone if I'm wrong on this, but this is what I feel is correct.
|
Kentor

United States5784 Posts
|
If you want to understand... read your chem book. If you want answers, go to yahoo answers. This is a pretty lame blog. Plus I can tell you that some of the answers you've been given are wrong.
|
On September 07 2008 07:48 letsbefree wrote:Hi guys, as a continuation from the last post, I firstly want to thank you all for helping me out. But apparently due to the inability of my chemistry teacher to teach, i have a few additional questions that i hope you guys can help you answer. Right now I'm studying electron configuration, and learning how to write elements using the 4 quantum numbers: n, L, ml, and ms. Plz refer to the below link for a moment: http://images.google.ca/imgres?imgurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads_files/image002.jpg&imgrefurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads.htm&h=412&w=609&sz=17&hl=zh-CN&start=1&um=1&usg=__3F_i4ommegoDLULRa80xkMvvEeQ=&tbnid=tIokQ6UbR59zTM:&tbnh=92&tbnw=136&prev=/images?q=spdf,+shape+of+orbital&ndsp=18&um=1&hl=zh-CN&sa=NThe shape of the orbitals of electrons are s, p, d, f, and so on. I know that at the first energy level, n = 1, thus L = 0, which means the shape of the orbital is "s" capable of holding 2 electrons in a spherical shape. When n = 2, L = 0 and 1, which means the orbitals at that energy level are in the shape of "s" and also "p". "s" can hold 2 electrons while "p" can hold 6, for a total of 8. But here comes my first question: do the orbitals "s" and "p" BOTH exist at the second energy level when n = 2, while the orbital at the first energy level "s" is separate from the orbitals in the second energy level?
Yes.
*In addition, in my chemistry notes the terms px, py, pz are also used. Do all three terms represent ONE "p" orbital?
Yes. x,y, and z, like the axes of a cartesian coordinate system. each hold two electrons. hence 6 total per p orbitals per energy level.
When n = 3, L = 0, 1, and 2. Which means the orbitals are "s", "p", and "d". "s" and "p" together can hold a total of 8 electrons. However, "d" supposedly can hold 10 electrons, and according to the link, the orbital "d" include 5 different orientations based on different x-y-z axises. My second question is, do all FIVE shapes exist together as the energy level "d"? If so, a total of 10 electrons in 5 orientations mean that each orientation can hold 2 electrons. How come there are 4 circles in each orientation then?
The circles aren't electrons; they are representing the probable distribution of the electrons within a given orbital in space around the nucleus. There are two electrons (max) in each d-orbital, and either can be found anywhere within that orbital (at that given probability, which is probably like 80%... technically an electron can be anywhere at any given moment, even across the universe if it felt like it).
There are five different d-orbitals for ten total, correct. So each energy level containing d-orbitals (and not f or higher) can have a maximum of 18 electrons.
Lastly, ml is the magnetic quantum number, which can equal to -L .... 0 ..... + L, and it tells you the spatial orientation of the orbital, whether its along the x-axis, y- axis, or z-axis. Which value of ml corresponds with each orientation??
never heard of this, sorry.
When it comes to the ms, which is the spin quantum number that tells the direction of spin of the electrons, for each orientation, DO I have to assign the positive 1/2 for the first electron first? Or does it not matter?
it does not technically matter; what matters is that two electrons in the same orbital must have complementary (aka opposite) spins. the positive/negative distinction is arbitrary in this case.
|
you seemed to have ignored our advice of reading your textbook... lol...
|
I concur, read the book. They should have good explanations.
In short:
1. Yes, the s orbitals are separate. Each contain 2 electrons.
2. There are 3 P orbitals that make up the entire P shell (one in the Px plane, one in Py plane, and one in the Pz plane). This is, of course, depends on how many shells you have. For example, neon only has one full P shell (6 electrons -- 2 in each px, py, pz), but Argon has 2 full P shells.
3a. There aren't 5 planes in which the D orbitals operate; that picture is pretty much BS and only shows one variation. They are weird ass shapes depending on equations dealing with heisenburg uncertainty and quantum mechanics and they represent the probability "cloud" where the electron(s) are at any given time. Don't worry about this. Just know there are 10 electrons and thus 5 orbitals for D (same with F with 7 orbitals and 14 total electrons can fill each).
3b. If you look at the periodic table closely, you'll see that that the group I and II series correspond to S shell (helium is S shell as well though); III, IV, V, VI VII, VII correspond to P shell 6 electrons in total; D is the transition metals, 10 wide in total, and F is the lanthanides and actinides.
4. Don't think you need to know which ml corresponds to which p axis. If I were to guess it would be X,Y,Z -1,0,1 heh..
5. Assign spins either up or down. It doesn't matter. There some rule I can't remember what it's called but it adds only up or only down spins to orbitals before you add the second. Therefore, nitrogen has one electron in px, py and pz respectively all in either the up or down configuration.
p.s. Read the damn text.
|
MY SCHOOL IS FUCKING CHEAP WE HAVE NO TEXTBOOK!
|
|
|
5) It either doesn't matter or you start with 1/2 and then go to -1/2 after all the +1/2 are taken (I advise you to be safe just in case and stick to using positive first that way you don't mess up)
everything eshlow said looks right.
|
ohhh god i don't feel like thinking right now, or i would help. just listen to bill, he's always right
(seriously)
|

United States24483 Posts
On September 07 2008 08:26 letsbefree wrote:
MY SCHOOL IS FUCKING CHEAP WE HAVE NO TEXTBOOK!
Don't blame your teacher for not teaching you if the school isn't providing a reasonable level of support lol
|
On September 07 2008 07:48 letsbefree wrote:Hi guys, as a continuation from the last post, I firstly want to thank you all for helping me out. But apparently due to the inability of my chemistry teacher to teach, i have a few additional questions that i hope you guys can help you answer. Right now I'm studying electron configuration, and learning how to write elements using the 4 quantum numbers: n, L, ml, and ms. Plz refer to the below link for a moment: http://images.google.ca/imgres?imgurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads_files/image002.jpg&imgrefurl=http://lincoln.pps.k12.or.us/lscheffler/OrbitalShapesOverheads.htm&h=412&w=609&sz=17&hl=zh-CN&start=1&um=1&usg=__3F_i4ommegoDLULRa80xkMvvEeQ=&tbnid=tIokQ6UbR59zTM:&tbnh=92&tbnw=136&prev=/images?q=spdf,+shape+of+orbital&ndsp=18&um=1&hl=zh-CN&sa=NThe shape of the orbitals of electrons are s, p, d, f, and so on. I know that at the first energy level, n = 1, thus L = 0, which means the shape of the orbital is "s" capable of holding 2 electrons in a spherical shape. When n = 2, L = 0 and 1, which means the orbitals at that energy level are in the shape of "s" and also "p". "s" can hold 2 electrons while "p" can hold 6, for a total of 8. But here comes my first question: do the orbitals "s" and "p" BOTH exist at the second energy level when n = 2, while the orbital at the first energy level "s" is separate from the orbitals in the second energy level?
yes, that is right.
*In addition, in my chemistry notes the terms px, py, pz are also used. Do all three terms represent ONE "p" orbital?
no, they represent 3 'p' orbitals. Each of the orbitals has 2 electrons in it.
When n = 3, L = 0, 1, and 2. Which means the orbitals are "s", "p", and "d". "s" and "p" together can hold a total of 8 electrons. However, "d" supposedly can hold 10 electrons, and according to the link, the orbital "d" include 5 different orientations based on different x-y-z axises. My second question is, do all FIVE shapes exist together as the energy level "d"? If so, a total of 10 electrons in 5 orientations mean that each orientation can hold 2 electrons. How come there are 4 circles in each orientation then?
The 4 'circles' you are talking about happen to be where the electrons would be found 95% of the time. At most 2 electrons would occupy all four lobes or 'circles' as you called them. 's' orbitals have one lobe that is a sphere and can hold two electrons. 'p' orbitals have 2 lobes and 2 electrons can occupy each orbital. This means the electrons could be found in either lobe at any time, with a 95% probability of course. 'd' orbitals have 4 lobes each. Each 'd' orbital can have 2 elecrons located in any of those 4 lobes. 'f'' orbitals have 8 lobes i believe, but maybe one or two of the 'f' orbitals might be differently shaped, i dont quite remember(its been a few years for me).
Lastly, ml is the magnetic quantum number, which can equal to -L .... 0 ..... + L, and it tells you the spatial orientation of the orbital, whether its along the x-axis, y- axis, or z-axis. Which value of ml corresponds with each orientation??
I dont think asking that question makes sense. Which axis is x,y, or z is completely arbitrary.
When it comes to the ms, which is the spin quantum number that tells the direction of spin of the electrons, for each orientation, DO I have to assign the positive 1/2 for the first electron first? Or does it not matter?
It is standard practice to assign the positive 1/2 first, but in reality 50% of the atoms would have a positive 1/2 and the other 50% have a negative 1/2.
I hope that helps. Im not sure if I explained it in the detail you need. I might be a little off on one or two things, but I did do 2nd year chemistry in university so hopefully I have remembered everything correctly. Quantum chemistry use to be my favourite part of chemisty.
|
On September 07 2008 08:32 Bill307 wrote:I think talismania covered this very well.  letsbefree: just ignore what Hippo says. I, for one, like these blogs: they help me recall all the stuff I learned in Chemistry class, at a minimum. I don't recall the "ml" either, unfortunately.
Just ignore what Bill307 said. Other people don't enjoy seeing questions about elementary level (although the topic itself is advanced) chemistry when there's a whole site dedicated to cheating on homework.
|
Since your question was more or less answered already I just wanted to give you the advice to buy a real text book.
It'll be 100-200 bucks but TL can only take you so far =P
If you are just starting chem Chemical Principles by Zumdahl works well.
|
|
|
|
|
|